
Calculate the efficiency of packing in case of a metal crystal for (i) simple cubic (ii) body- centred cubic (iii) face - centred cubic . (With the assumptions that atoms are touching
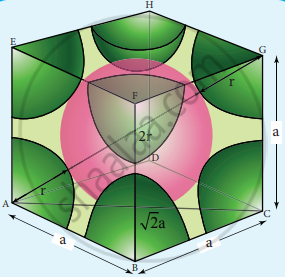
Calculate the percentage efficiency of packing in the case of body centered cubic crystal. - Chemistry | Shaalaa.com
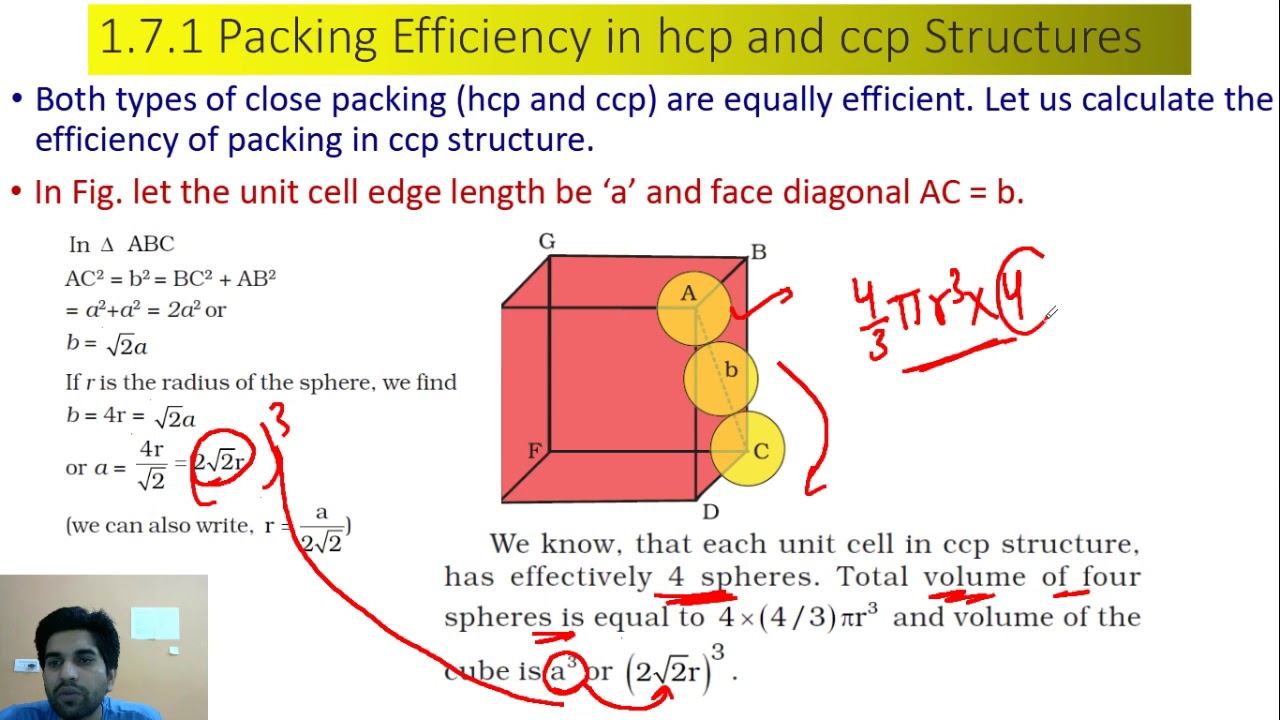
packing efficiency ncert class 12 , packing efficiency in FCC, BCC, Primitive unit cells | chemistry - YouTube















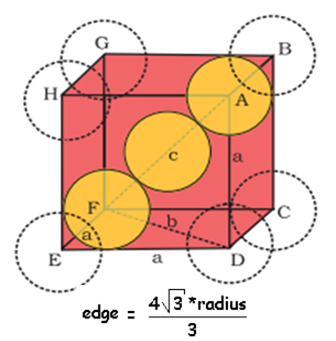
![Kannada] Calculate the number of particles in Body Centered Cubic (BC Kannada] Calculate the number of particles in Body Centered Cubic (BC](https://d10lpgp6xz60nq.cloudfront.net/physics_images/SPH_CHE_HND_BOK_PUC_JUL_15_E01_036_S01.png)


