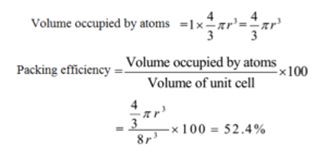Three-dimensional packing of spheres: bcc (Im 3m), fcc (Fm 3m) and hcp... | Download Scientific Diagram

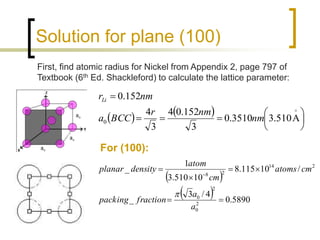
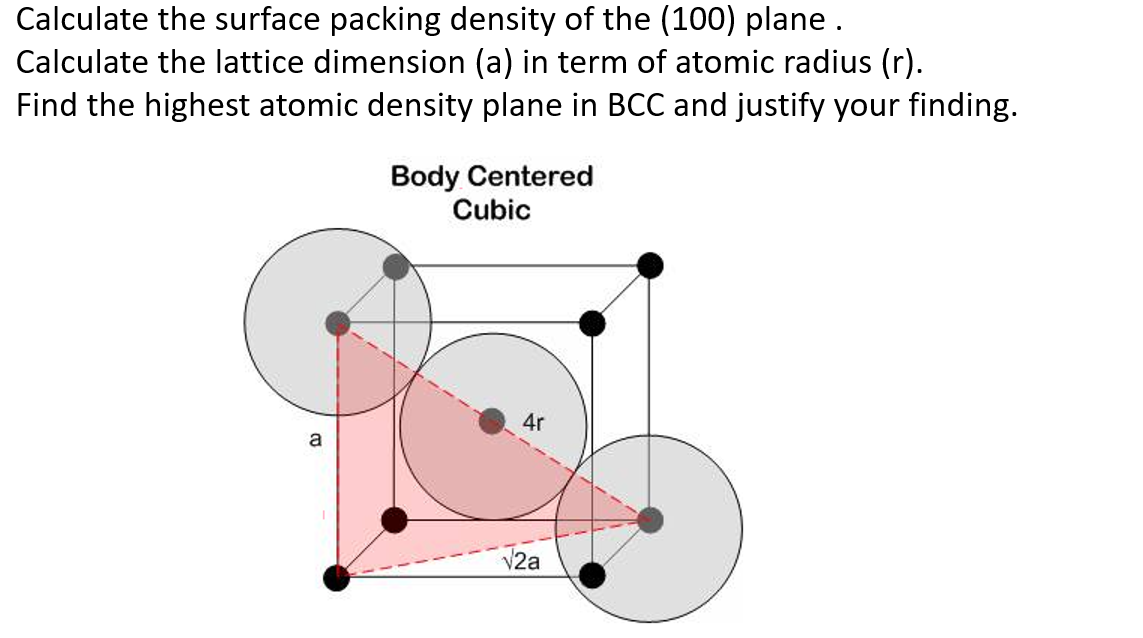
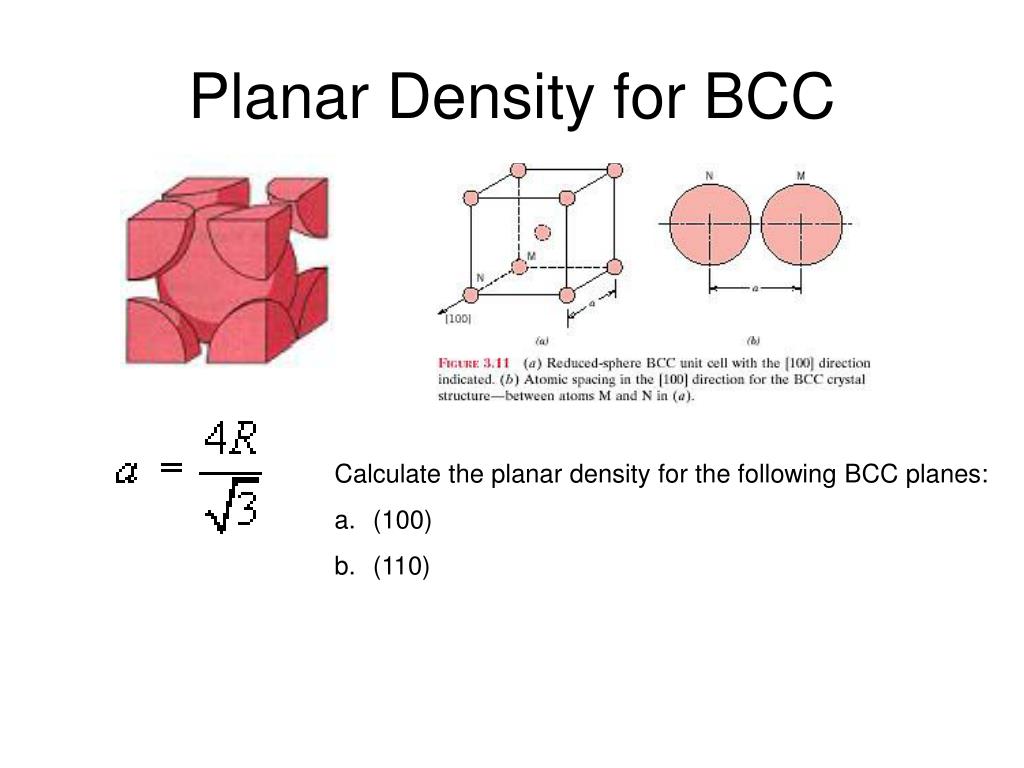
Determine the planar density and packing fraction for BCC lithium in the (100), (110), and (111) planes. Which, if any, of these planes is close packed? | Homework.Study.com

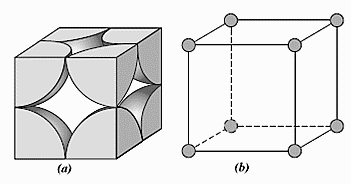
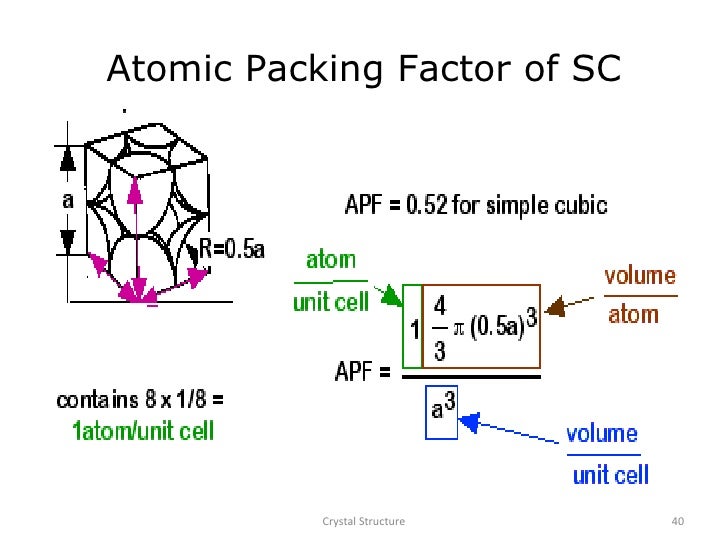
SOLVED: 1. Calculate the atomic packing density of the following structures: i. Simple cubic ii. Face-centered cubic iii. Body-centered cubic iv. Hexagonal close packing 2. The density of BCC iron is 8,000

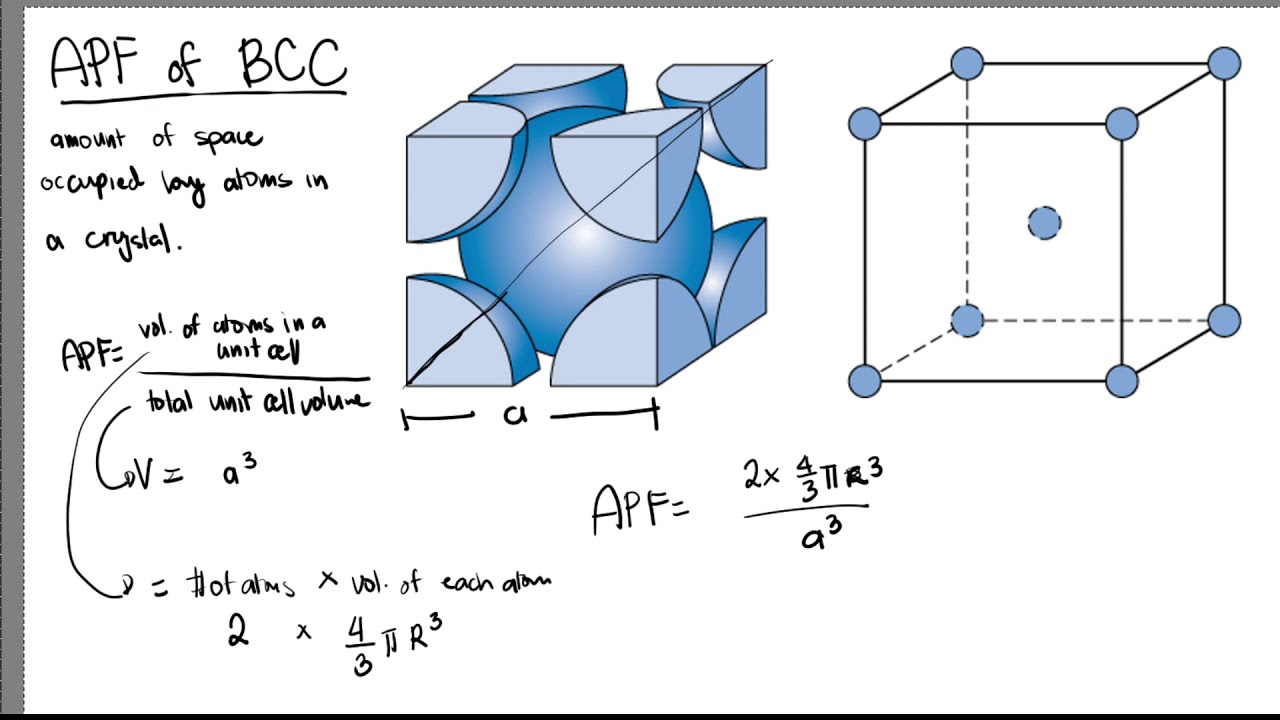
How can i calculate packing efficiency of bcc structure - Chemistry - The Solid State - 13423615 | Meritnation.com